Vocabulary
Chemical Bond- A attraction between atoms that allow the formation of chemical substance that contain two or more atoms.
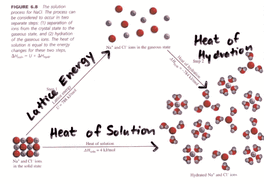
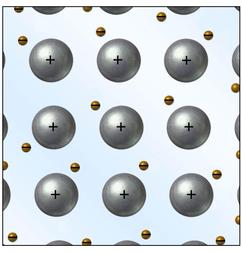
Cation- A cation is an ionic species with a positive charge. Anion- An ionic species that have a negative charge. Ionic bond- A chemical link between two atoms caused by the electrostatic force between oppositely-charged ions in an ionic compounds. Electrolyte- A substance which forms ions in an aqueous solution. Lattice energy- The process in which oppositely charged ions in the gas combine to form an ionic lattice in the solid phase. Metallic Bond -The reaction between molecules within metals called alkali reactive force. Formula unit-The empirical formula of ionic or covalent network solid compound used as an independent entity for chronometric calculations. Monatomic ion- A ion consisting of one or more atoms of a single element. Oxidation number-The number of electrons lost, gained, or shared as a result of chemical bonding. Polyatomic ion- Known as molecular ion, is a charged ion composed of two or more atoms covalently bonded or of a metal complex. Oxonian- An anion containing one or more oxygen atoms bonded to another element. Electron Sea Model- A model of metallic bonding in which cations are considered to be fixed points within a mobile sea of electrons. Delocalized electrons- Electrons in a molecule, ion or solid metal that are not associated with a single atom or one covalent bond. |